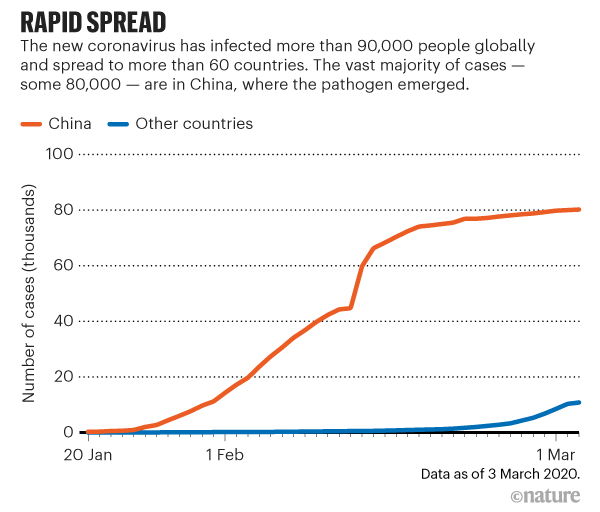
The team will share the results of their study at the SC21 conference. These simulations helped fill in the blanks the cryo-electron microscopy couldn’t capture and reconstruct the motion that we couldn’t otherwise understand to reach a biophysically meaningful interpretation.” “Everything had to come together in just the right place in just the right way, like an assembly line. “By coordinating this work across these sites, we could use all the strengths of the best state-of-the-art computing to perform these simulations,” Ramanathan said. The workflow builds on a strategy employed by Ramanathan and Rommie Amaro, a professor and endowed chair of chemistry and biochemistry at the University of California San Diego, to simulate the behavior of the virus’s spike protein, a study that won last year’s Gordon Bell Special Prize for COVID-19 research. With access to the ALCF AI Testbed, the team used a Cerebras wafer-scale engine to train deep learning models that were coupled with the various supercomputing systems. The team used a hierarchical artificial intelligence ( AI) framework running on Balsam, a distributed workflow engine across four of the nation’s top supercomputing systems - Summit, Oak Ridge Leadership Computing Facility’s ( OLCF) 200-petaflop flagship computer Theta, Argonne Leadership Computing Facility’s ( ALCF) 15.6-petaflop system Perlmutter, the National Energy Research Scientific Computing Center’s ( NERSC’s) 64.6-petaflop system and Longhorn, a subsystem of the Texas Advanced Computing Center’s 23.5-petaflop Frontera system - to simulate the process. 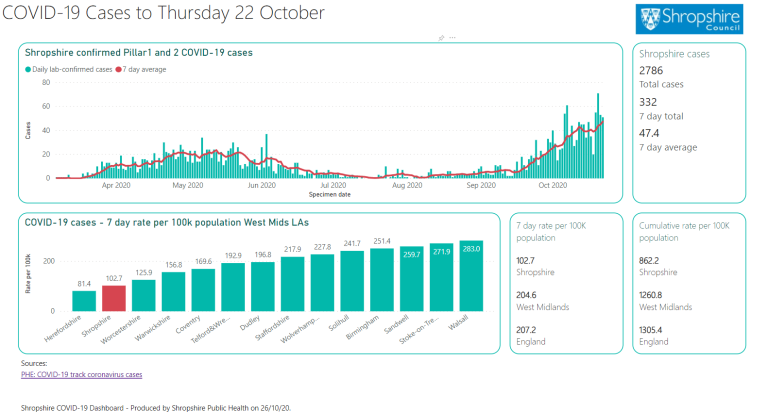 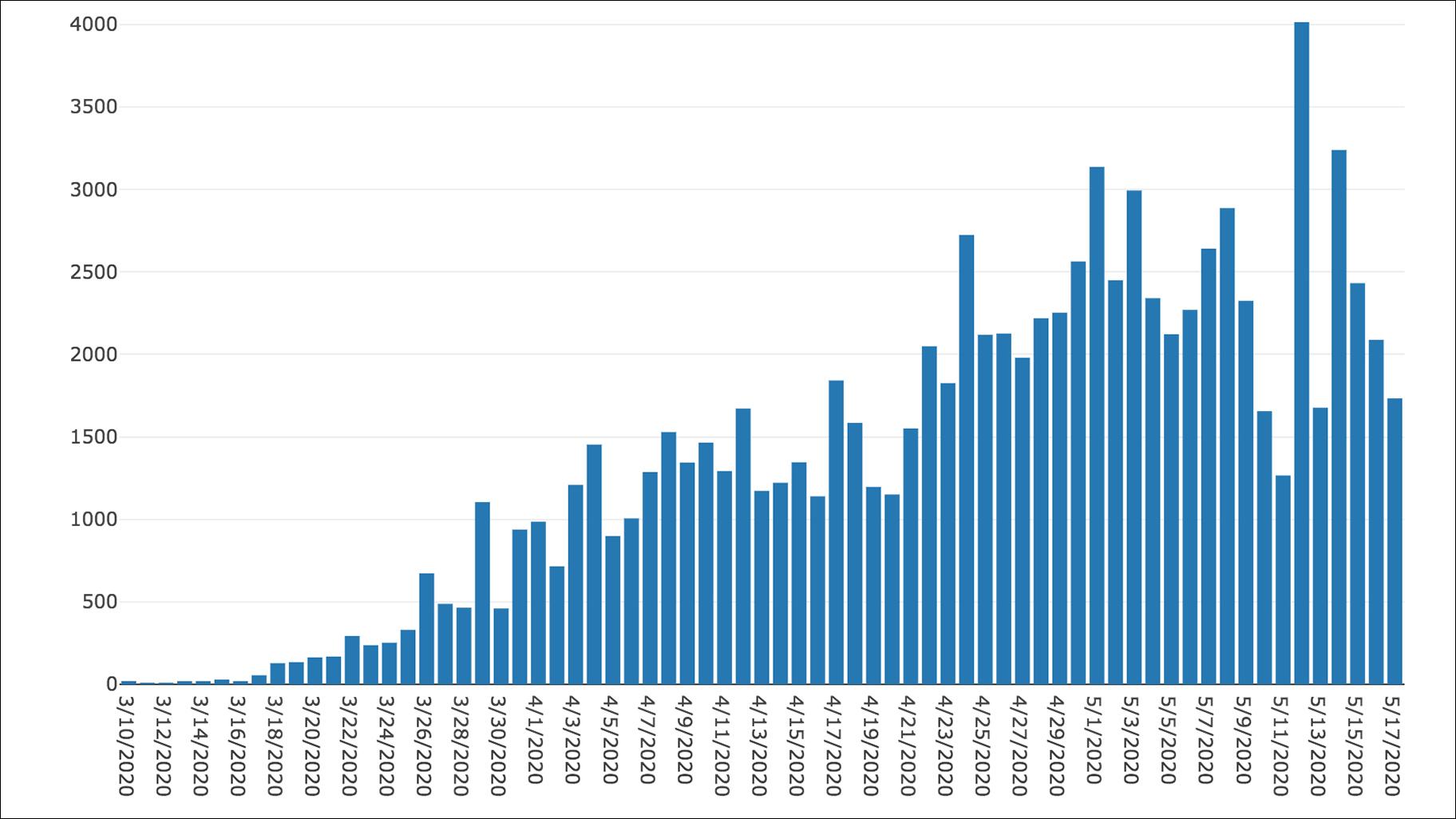
We needed to see that waltz and the machinery in motion in order to understand how to gum up this Swiss watch.” 
“The molecules move in a complicated pattern, like a waltz. “It’s critical to be able to see the interactions between the nonstructural proteins (which are mostly enzymes) as they process the viral RNA one base at a time,” Ramanathan said. But static images alone wouldn’t be enough to capture the workings of the copying process. The team used data from cryo-electron microscopy, a technique that flash-freezes molecules and pounds them with electrons to create 3D images, to take a closer look at the molecular machinery. “A number of scientists have done tremendous work to understand some of these individual parts, but nobody had looked at this complex from a broader view to try to understand how they all work together.” “It’s a system of roughly 2 million atoms, and there’s no single way to get a really good look inside,” Ramanathan said. The process essentially transcribes the ribonucleic acid, or RNA, that contains the genetic code for the virus, packages the RNA and pumps out the photocopies of itself to overwhelm the host cells. The coronavirus uses a precisely coordinated process known as the replication-transcription complex to reproduce at high speed when it invades a host’s cells. The prize will be presented at this year’s SC21 conference, Nov. The study earned the multi-institutional team a finalist nomination for the Association of Computing Machinery ( ACM) Gordon Bell Special Prize for High Performance Computing–Based COVID-19 Research. But first we had to better understand it.” “If we could find ways to block or gum up the copying process, we could discover new drugs to attack the virus. Department of Energy’s ( DOE) Argonne National Laboratory and the study’s lead author. “Think of it as like a Swiss watch, with precisely organized enzymes and nanomachines that come together like tiny gears to perform this function,” said Arvind Ramanathan, a computational scientist at the U.S. To find weapons to fight the coronavirus, scientists used the nation’s fastest supercomputer to peer inside the intricacies of how the virus reproduces itself.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
